Frequently diagnosed ECG conditions in clinical practice
Welcome to the guide on the most commonly conditions diagnosed by means of the ECG. We will examine a range of cardiac conditions, looking into their causes and symptoms and providing illustrations of how they can manifest on the ECG. Pacemaker detection is also discussed.
Please note that the conditions below are presented in simplified terms and illustrations for educational purposes only. A diagnosis may only be made by a cardiologist.
In this blog you will learn:
Acute coronary syndromes
Myocardial infarction
Unstable angina pectoris
Pericarditis and myocarditis
Atrial fibrillation
Brugada syndrome
Left and right ventricular hypertrophy
Left ventricular hypertrophy
Right ventricular hypertrophy
Ventricular tachycardia
Conduction disturbances
Atrioventricular blocks
Left and right bundle branch blocks
Pacemaker detection

Dive into our comprehensive e-book to explore:
- How the ECG works
- How to conduct a resting ECG measurement
- How to interpret the ECG
- Frequently diagnosed conditions
- Automatic interpretation
- A comparison of different resting ECG machines
- Technical specifications of the MESI mTABLET ECG
Acute coronary syndromes
This is a group of diseases where the blood flow to the heart decreases. They include myocardial infarction (STEMI and NSTEMI) and unstable angina pectoris.
Myocardial infarction
Myocardial infarction (MI) or colloquially ‘heart attack’ is caused by decreased or completely stopped blood flow to a part of the myocardium (i.e. the heart muscle, responsible for contracting the heart). If the coronary artery is blocked, the myocardium does not receive oxygen. This can lead to the death and necrosis of the myocardial cells. [3] [4]
Myocardial infarction can be ‘silent’ and go unnoticed, but in the worst case, it can cause hemodynamic deterioration (abnormal heart rate and blood pressure) and sudden death. Patients can feel chest discomfort or pressure radiating towards the arm, jaw, shoulder or neck.
MI shows dynamic changes on the ECG. Even if the initial ECG is non-diagnostic, serial ECG monitoring (i.e. for a period of time) can lead to the diagnosis.
There are two types of myocardial infarction – STEMI and NSTEMI. STEMI shows ST elevation on the ECG, but NSTEMI may show no ECG changes. Both can cause death of heart muscle tissue.
STEMI myocardial infarction
STEMI occurs due to complete and prolonged blockage of one or more coronary arteries. This causes transmural myocardial ischemia (a lack of blood supply that affects the full thickness of the myocardium). The result is injury or necrosis of the myocardium. [5]
STEMI occurs due to complete and prolonged blockage of one or more coronary arteries. This causes transmural myocardial ischemia (a lack of blood supply that affects the full thickness of the myocardium). The result is injury or necrosis of the myocardium.
The risk factors for STEMI include:
diabetes,
smoking,
dyslipidemia,
coronary artery disease (CAD) and
family history of CAD.
A typical sign is chest pain or discomfort, especially with acute STEMI. The symptoms are more severe compared to NSTEMI and unstable angina because a larger part of the myocardium becomes ischemic.
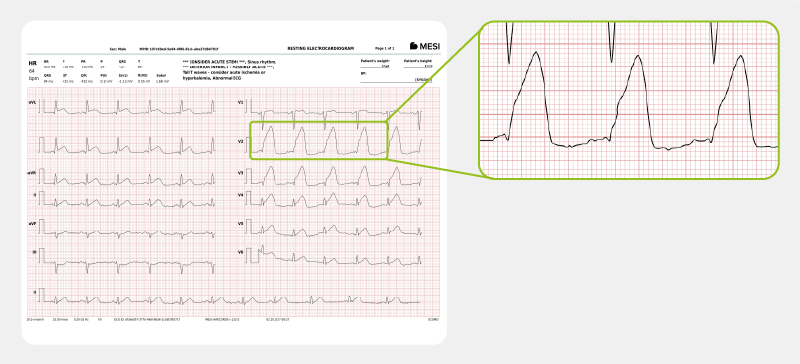
In the ECG, STEMI is characterised by ST elevation. After the occlusion of the coronary artery, ventricular fibrillation and ventricular tachycardia can follow.
For an actual case of STEMI myocardial infarction, click here.
NSTEMI myocardial infarction
NSTEMI is caused by:
severe coronary artery narrowing,
temporary occlusion, or
microembolisation of atheroma (fatty substance in arteries) and/or
thrombus (blood clot). [5]
A patient may be clinically unstable (e.g. shock, low blood pressure, left ventricular failure). The most frequent sign is chest pain, but less pronounced than in STEMI because the artery occlusion is partial.
NSTEMI does not show ST elevation, but there is an elevation of cardiac biomarkers (troponins) just like with STEMI. The ECG can show ischaemic changes, e.g. T wave changes, ST depression, or transient ST elevation; however, it can also show non-specific changes or be normal.
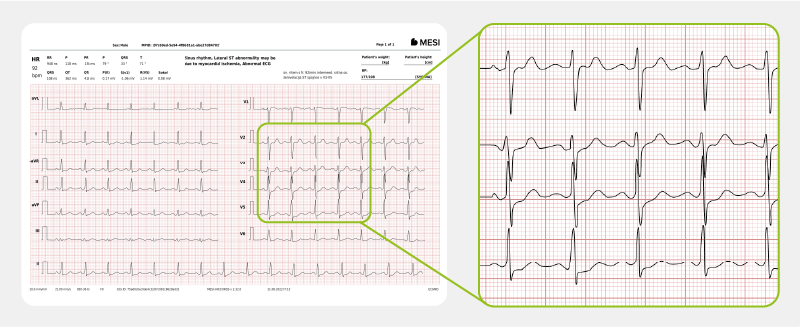
For an actual case of NSTEMI myocardial infarction, click here.
Unstable angina pectoris
Unstable angina pectoris is chest discomfort or pain caused by an insufficient flow of blood and oxygen to the heart. It can result in myocardial infarction. [6]
This condition is caused by coronary atherosclerotic disease – a plaque buildup in the coronary arteries leading to a thrombus that partially blocks the blood flow. Less frequently, it is caused by vasospasm of a coronary artery.
Risk factors include:
diabetes,
hypertension,
family history,
obesity,
chronic kidney disease,
anemia,
autoimmune disorders,
smoking (current or former) and
substance abuse (cocaine, amphetamines).
In difference to MI, unstable angina pectoris shows normal troponin levels.
The symptoms are shared with coronary atherosclerotic disease. The most frequent symptom is shortness of breath and chest pain, but weaker than in STEMI because of partial coronary artery occlusion. The pain or discomfort may radiate to either arm or the jaw. Vomiting, nausea, dizziness, diaphoresis and palpitations can also occur. The pain may worsen with physical exertion and diminish with rest.
The ECG in unstable angina can show flattened T-waves, hyperacute T-wave, inverted T-waves, and ST depression. ST elevations indicate STEMI. There is sinus rhythm in difference to an infarction.
Pericarditis and myocarditis
These two conditions are inflammations in different parts of the heart. They are difficult to distinguish because they are often present together.
Pericarditis
This is an inflammation of the pericardium (the sac that contains the heart and the roots of the major vessels). Most often, pericarditis is caused by (viral) infection. For this reason, it can affect all ages.
It can also have many systemic and local causes:
post-infarction,
acute myocardial infarction,
rheumatoid arthritis,
lupus,
tuberculosis,
post cardiac surgery (haemorrhagic pericarditis), etc.
The most frequent symptom of pericarditis is chest pain, which is difficult to differentiate from acute MI. The pain can be strong and accompanied by cold sweats, anxiety and/or tachycardia.
Myocarditis
An inflammation of the myocardium and the interstitium (the connective space between the cells in the myocardial tissue).
It can be caused by:
viral or bacterial infection,
auto-immune reactions,
fungi,
parasites,
borreliosis and
HIV/AIDS.
Patients show abnormal heart rate, faint chest pain and progressive heart failure.
On the ECG, acute pericarditis and myocarditis are characterised by non-specific ST segment changes. There can also be irregularities in ventricular and supraventricular rhythm, and T wave abnormalities.
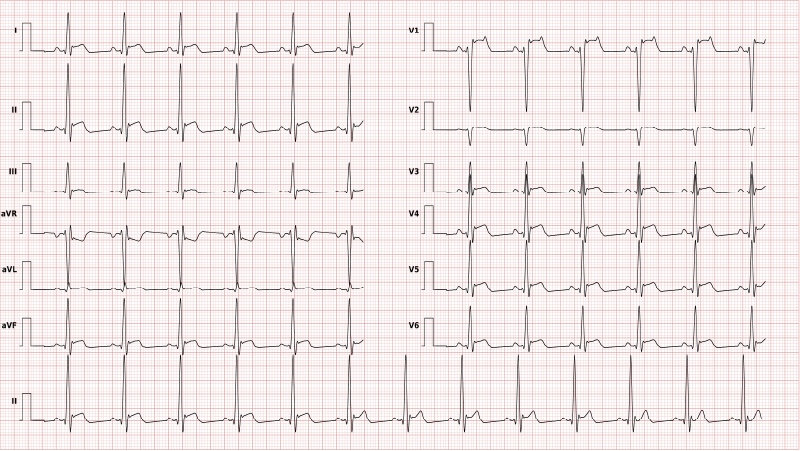
Atrial fibrillation
Atrial fibrillation is the most common pathologic tachyarrhythmia (abnormal heart rhythms with a heart rate of 100+ beats per minute). The condition suddenly occurs in the heart atria.
Common ECG signs of atrial fibrillation are irregular ventricular rate and absence of P waves.
The most common symptoms are (a combination of) fatigue, discomfort in the throat and the chest, palpitation, dyspnoea and diminished exercise ability. Approximately one fourth of patients do not have symptoms; in this case, the condition is detected by means of the ECG.
The risk for this condition increases with age. It occurs in less than 1% of people between 60 and 65, but in 8-10% people older than 80.
The risk is also increased by:
hypertension,
sleep-related breathing disorders,
heart valve disease and
congestive heart failure.
Common ECG signs of atrial fibrillation are irregular ventricular rate and absence of P waves.
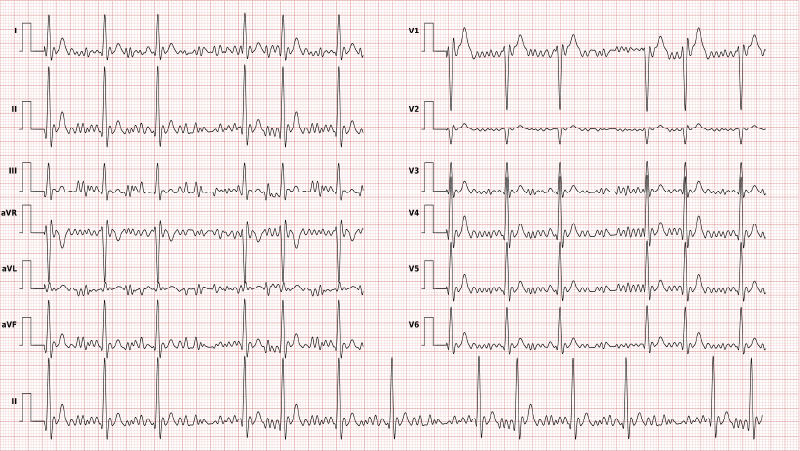
Brugada syndrome
Brugada syndrome (BrS) is a rare inherited arrhythmia condition. It can lead to life-threatening ventricular fibrillation and sudden cardiac death, without identifiable structural abnormalities. [8]
It generally affects middle-aged patients (45 at diagnosis), with an 8 times higher diagnosed occurrence in men.
Risk factors for Brugada syndrome are:
family history of this condition,
race (most frequent in Asians),
fever (it doesn’t cause the condition, but it can irritate the heart and therefore be a trigger), and
the use of certain prescription medications or cocaine.
Many patients with BrS are asymptomatic. Syncope (fainting due to abnormal blood flow to the brain) or (aborted) sudden cardiac death are often the first symptoms. They frequently appear at night or at rest.
The ECG shows a specific but labile pattern, defined as a ≥ 0.2 mV ST-segment elevation in the precordial leads (V1-V3). The ECG can also be ‘silent’ (non-specific) and sodium blockers are applied for the condition to show.
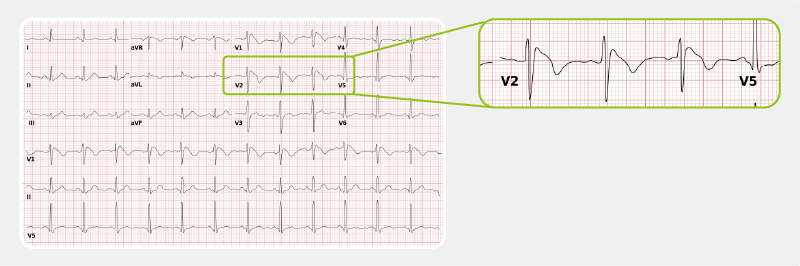
Left and right ventricular hypertrophy
In hypertrophy, the heart muscle becomes thicker due to various causes. Left and right ventricular hypertrophy can be distinguished on the ECG. [9] [10]
Left ventricular hypertrophy
Left ventricular hypertrophy (LVH) is caused by increased left ventricular workload, e.g. due to aortic valve stenosis or hypertension.
LVH may be present without symptoms for years. As it worsens, fatigue, shortness of breath, heart palpitations, chest pain (especially during exercise), dizziness or fainting can appear.
Due to the left ventricular wall thickening, the QRS complexes become larger, especially in leads V1-V6. There is a deep S wave in V1 and a high R wave in V4. Some ST depression is often observed in leads V5-V6 (so-called ‘strain pattern’).
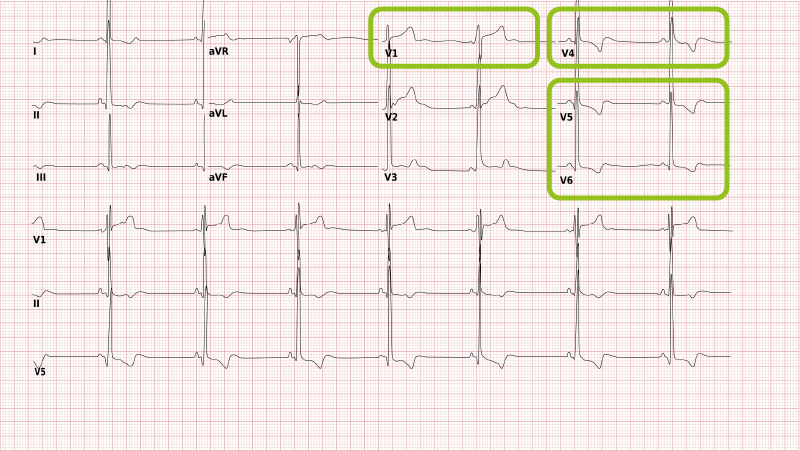
Right ventricular hypertrophy
Right ventricular hypertrophy (RVH) is caused by increased right ventricular workload, e.g. due to pulmonary embolisation or emphysema.
RVH occurs due to congenital heart disease or lung disease. The condition itself may be without symptoms, but there may be some due to the associated heart or lung problems.
The ECG shows a negative QRS complex in I (and therefore a right heart axis) as well as positive QRS complex in V1.
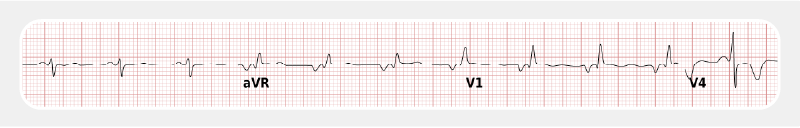
Ventricular tachycardia
Ventricular tachycardia (VT) is a potentially fatal rhythm disorder (arrhythmia) in the heart ventricles. [11]
The clinical manifestations and consequences of VT are diverse:
ventricular fibrillation,
syncope,
electrical storm,
cardiogenic shock,
cardiac arrest and
sudden cardiac death.
VT frequently occurs due to:
structural heart disease (conditions that affect the muscles, chambers, walls or valves of the heart),
channelopathies (i.e. Brugada syndrome),
ischemia,
electrolyte disturbances, and
around old scar tissue in the heart, e.g. after myocardial infarction.
VT is a sequence of three or more ventricular beats. The frequency is higher than 100 bpm, mostly between 110 and 250 bpm. The chief management strategy for VT are implantable cardioverter-defibrillators (ICDs). They reduce the number of death cases, but not VT recurrences.
The conditions described in the continuation are those connected with conduction disturbances. They are perhaps less well-known, but are just as frequent as the ones above and have characteristic ECG patterns.
Conduction disturbances
Conduction disturbances or heart blocks are problems with the cardiac conduction system, which controls the heart rate and rhythm.
Atrioventricular blocks
The conduction between the atria and the ventricles is disturbed. [12]
Dependent on the severity, the following symptoms can appear:
chest pain,
fatigue,
dyspnea,
(pre)syncope,
sudden cardiac arrest.
Atrioventricular blocks lead to an increased PQ interval or to P waves (i.e. atrial activity) that are not (immediately) followed by QRS complexes (i.e. ventricular activity). This can be clearly seen on the ECG.
There are three degrees of atrioventricular block. They differ in severity.
FIRST-DEGREE AV BLOCK
This degree is relatively harmless because the QRS complex is late, but not absent. Some people are born with this condition and can lead normal lives if it does not aggravate. Other causes are:
inferior myocardial infarction,
myocarditis,
increased vagal tone (e.g. in athletes),
hyperkalemia,
post cardiac surgery and medications (e.g. beta-blockers), and
advanced age due to the degeneration of the conduction system.
First-degree AV block is marked by prolonged PQ (> 0.20 sec), followed by the QRS complex.
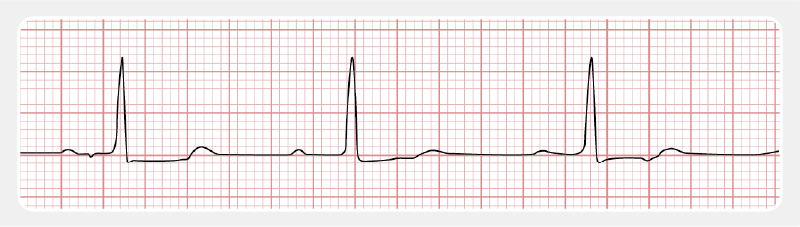
SECOND-DEGREE AV BLOCK MOBITZ TYPE 2
The heart beats are dropped irregularly. Hemodynamic instability or bradycardia can occur at any time, resulting in syncope or even sudden cardiac death. A pacemaker is required.
Some of the causes are:
anterior MI resulting in septal infarction and necrosis;
idiopathic fibrosis of the conduction system;
inflammations (e.g. Lyme disease, myocarditis);
autoimmune conditions (e.g. lupus, sclerosis);
electrolyte imbalance (e.g. hyperkalemia);
post cardiac surgery (e.g. mitral valve repair);
medications (e.g. non-dihydropyridine calcium channel blockers, beta-blockers).
There is no PQ prolongation in Mobitz type 2 AV block. There is a risk of worsening to a complete heart block.
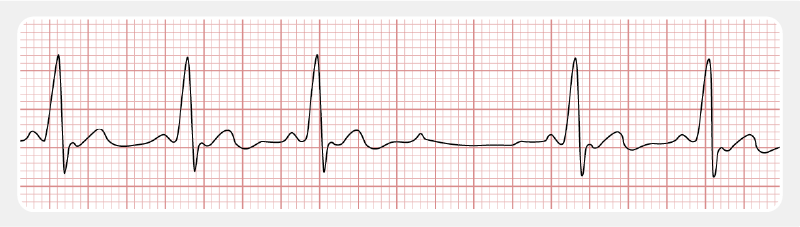
THIRD-DEGREE AV BLOCK
The block is total – there is no atrioventricular conduction. The blood supply to the brain can be insufficient, which results in loss of consciousness.
Absent ventricular rhythm results in asystole and death. The causes are often the same as in second degree AV block, including the degeneration of the conduction system due to advanced age.
A permanent pacemaker is required.
On the ECG, there is no connection between P waves and the QRS complexes.
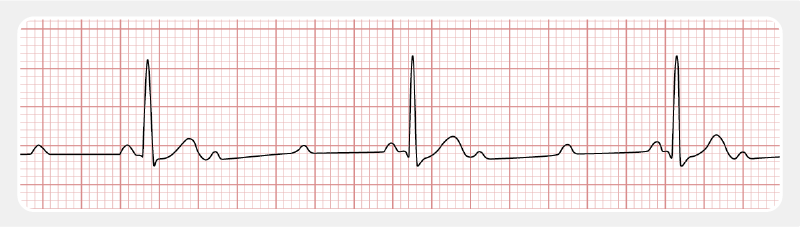
Left and right bundle branch blocks
LEFT BUNDLE BRANCH BLOCK (LBBB)
In LBBB, the conduction in the left bundle is slow. [13] [14]
Some factors are:
hypertension,
myocarditis,
ventricular hypertrophy,
ischemic heart disease and
heart failure.
Major causes are ischemia and infarction; LBBB may also imitate or mask them. Therefore, the management of LBBB is similar to that of acute STEMI.
LBBB is asymptomatic, but there is a characteristic ECG pattern. The conduction is slow, causing delayed depolarisation of the left ventricle. Due to slower depolarisation of the left ventricle, there is a significant negative QRS in V1-V3.
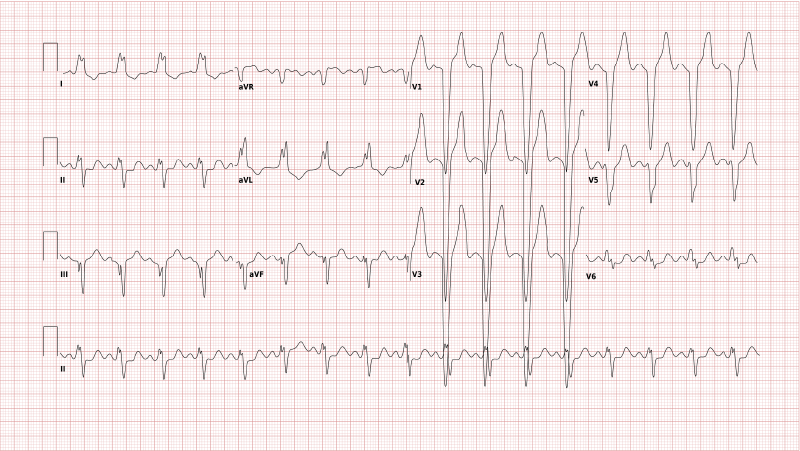
RIGHT BUNDLE BRANCH BLOCK (RBBB)
The conduction in the right bundle is slow. There can be a delay or a partial obstruction on the path of the electrical impulses to the right ventricle (‘incomplete RBBB’) or a full obstruction (‘complete RBBB’).
Some people are born with incomplete RBBB and can lead normal lives. It is also caused by other conditions such as:
hypertension,
structural cardiac disorders (e.g. myocarditis, cardiomyopathy, ischemic heart disease),
Brugada syndrome or
pulmonary hypertension.
RBBB is mostly asymptomatic and is often accidentally detected at routine ECGs. Occasionally, fainting (presyncope) may occur and should be investigated because serious block events can follow.
The depolarisation of the right ventricle is delayed, so the electrical activity in the left ventricle is mostly finished by that time. The last electrical activity can thus be seen to the right, with a markedly positive QRS complex in V1.
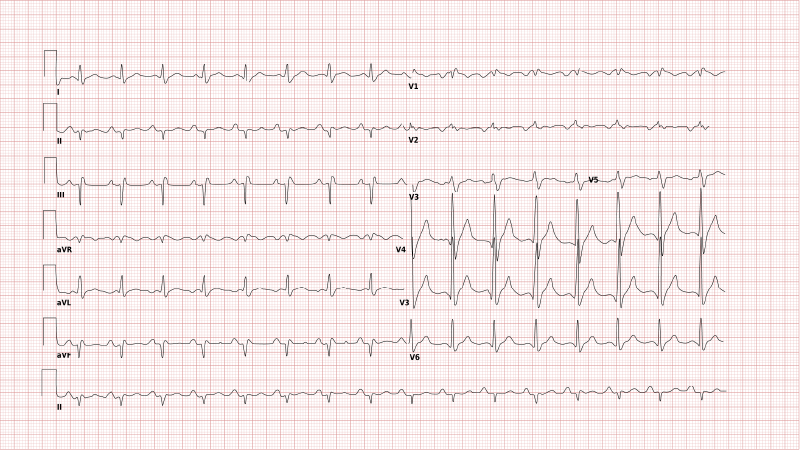
Pacemaker detection
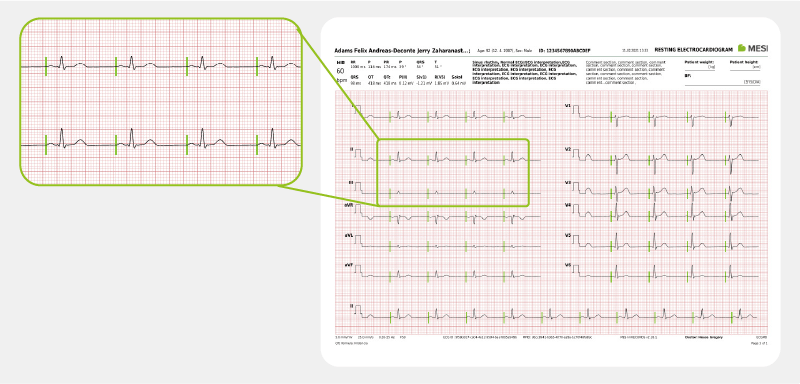
A pacemaker is implanted when electrical impulse conduction in the heart is dangerously disturbed due to abnormal heart rhythms that make the heart miss beats or beat too slowly. [16]
The pacemaker rhythm can be detected on the ECG. It is shown as pacemaker spikes – vertical signals that represent the electrical stimulation activity of the pacemaker.