Utility of Toe-Brachial Index (TBI) for diagnosis of PAD
The negative effects of many risk factors for peripheral artery disease (PAD) are not only limited to the patient’s well-being but can interfere or even prevent timely diagnosis of asymptomatic patients. We are talking about medical conditions such as diabetes, which also affects the severity and progress of PAD. They are the most prominent causes of incompressible arteries in the lower extremities and associated difficulties in diagnosing PAD using standard methods.
In this blog you will learn:
Why use the Toe-Brachial Index (TBI) for peripheral arterial disease (PAD) diagnosis?
What are the differences between Ankle-Brachial Index (ABI) and Toe-Brachial Index (TBI)?
Is Toe-Brachial Index (TBI) useful in designing a treatment programme for diabetic gangrene?
Why use the Toe-Brachial Index (TBI) for peripheral arterial disease (PAD) diagnosis?
There are several established methods for diagnosing PAD, differing in their accuracy, reliability, and cost, but none as convenient and cost-effective as ABI measurement (1, 2, 3). The ABI is convenient due to its non-invasive nature, while the cost-effectiveness derives from the relatively simple measurement procedure, particularly if the measurement is performed using an oscillometric-plethysmographic device (4, 5, 6). However, it has one major flaw, which comes to light in specific situations – its diagnostically nearly useless results in patients with incompressible arteries.
Under the term incompressible (calcified or stiff) arteries we mean arteries affected by medial arterial calcification (also known as Mönckeberg’s sclerosis) (7). They require higher cuff pressures to achieve compression; that leads to false ABI results, which are most common in patients with diabetes, renal insufficiency, and rheumatoid arthritis (8, 9, 10, 11).
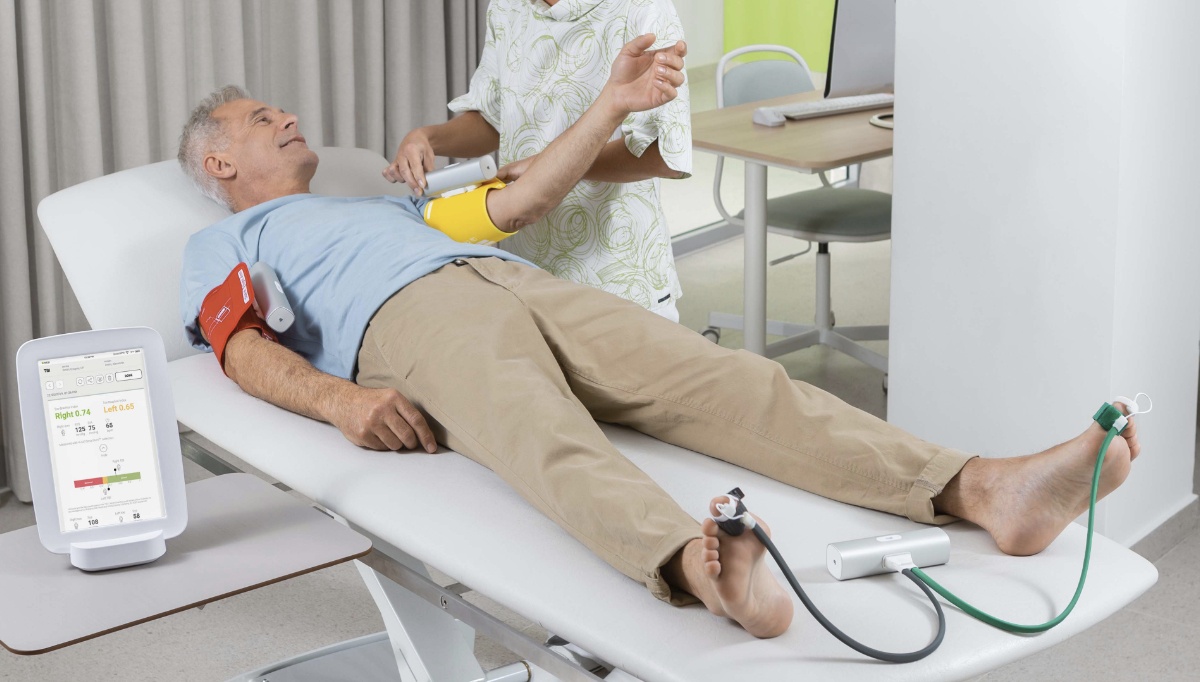
High ABI (≥1.30 or 1.40) has some limited diagnostic value, such as increased risk for myocardial infarction (MI), but offers no information about whether the examined patient has PAD and how severe it is (12). Fortunately, calcification rarely affects the arteries in toes, making them suitable for measuring blood pressure (13). However, in this case, the measurement is called the Toe-Brachial Index (TBI).
What are the differences between Ankle-Brachial Index (ABI) and Toe-Brachial Index (TBI)?
Apart from the name and the site where the blood pressure is measured, the most obvious difference between the two diagnostic methods is the diagnostic equipment used and the numerical values calculated from measurements. The pressure cuffs are considerably smaller and have an added photoplethysmograph sensor for detection of blood volume changes. Numerical values are likewise lower than those in ABI: they usually range from 0.7 upwards for healthy individuals, while a TBI <0.70 is generally indicative of PAD (14). However, there are similarities with ABI in the association of lower TBI values with overall general and cardiovascular health.
Studies have shown that low TBI is associated with increased risk of recurrent CVD and progression of diabetic nephropathy in patients with type 2 diabetes and is even useful as an informative predictor of mortality in dialysis patients (15, 16, 17). TBI is, in general, particularly suited for diagnosing PAD in patients with end-stage renal disease (ESRD) (18). Many of those patients are also battling painful ulceration (contraindicative for an ABI measurement) as are those with other conditions like diabetes, which has many serious complications like diabetic gangrene (19). Like nearly all vascular conditions that threaten limb viability, it is difficult to treat, particularly if the patient has an underlying PAD. Designing a proper treatment regime for such patients can be challenging but can also be made easier with the use of TBI.
Is Toe-Brachial Index (TBI) useful in designing a treatment programme for diabetic gangrene?
The association of PAD with diabetes mellitus and the mutually destructive synergy is well researched and does not need additional introduction. However, few studies have touched upon the usability of cost-effective diagnostic methods for PAD like ABI and TBI in designing and guiding treatment regimens for the treatment of diabetic complications like gangrene. One of those few is a study conducted in Korea that consisted of 15 patients (12 males and 3 females, altogether 30 limbs) with diabetic gangrene or intermittent claudication to assess the utility of TBI in assessing PAD in the aforementioned patients (20).
Researchers found TBI to be the method of choice for evaluating lower limb perfusion disorders and proved to be superior to ABI. Even more, some patients had a normal ABI score, but a low TBI score and consequently, PAD as confirmed by angiography (20). Had the TBI measurement not been performed, they would have been classified as PAD-free and not have received proper treatment. Out of eight of them, two had to have a skin graft and one had wound revision, while the rest had to have an amputation of one or two joints (20).
Normal ABI is, therefore, not the best indication of PAD status, particularly in patients who are likely to have incompressible arteries. They should be followed-up by a TBI measurement to confirm or deny any suspicions, or referred to a vascular specialist for additional diagnostics. Consecutive measuring of ABI and TBI also has other benefits, as noted by researchers. They have developed a treatment algorithm that enables faster detection of PAD in patients with diabetic gangrene.
First, a patient’s ABI is measured and, if it is below 0.9, the patient is referred to a vascular specialist for treatment. If the ABI score is between 0.9 and 1.3 (normal values) or more than 1.3 (incompressible arteries), a TBI measurement is performed. A TBI of at least 0.6 or more indicates no significant perfusion issues and that the patient requires only wound management. In contrast, a TBI of less 0.6 indicates angioplasty as a prerequisite to wound management.
The TBI is the only suitable alternative to ABI of comparable cost-effectiveness and convenience in patients with incompressible arteries and has applications beyond diagnosing PAD.
Provide complete 1-minute arterial assessment to your patients with advanced diagnostic ABI and TBI devices. LEARN MORE